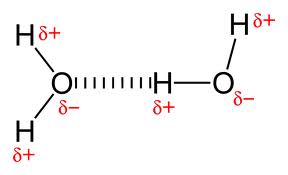
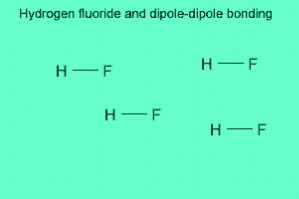
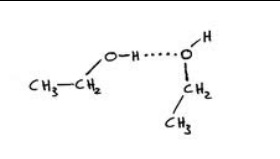
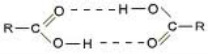Hydrogen Bonding occurs within molecules having a net dipolethat must be comprised of hydrogen that is in a covalent bond with either Fluorine (F), Oxygen (O), or Nitrogen (N).
|
THE STRONGEST FORCE...Hydrogen bonds are stronger than dispersion forces and dipole-dipole forces, but weaker than ionic and covalent bonds.
-A hydrogen atom's nucleus is very small &contains 1 proton. -Fluorine, oxygen and nitrogen are highly electronegative. So the electrostatic attraction is greater due to the huge difference in electronegativity. |