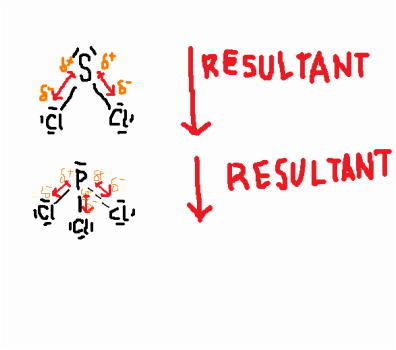
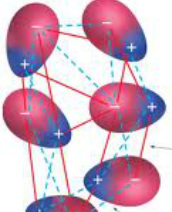
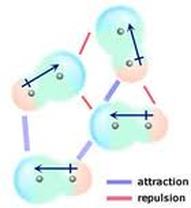
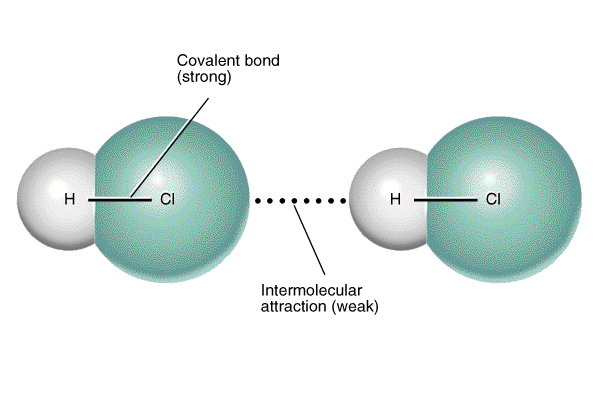
Dipole-Dipole Forces only occur within polar molecules-In a polar molecule, one end is more electronegative than the other, so this end is partially negatively charged, and the other end is partially positively charged.
-This creates a dipole. -The positive region of each molecule is attracted to the negative region of its neighboring molecules. -Unlike London Dispersion Forces, the net dipole is permanent. -Dipole-Dipole Forces occur within SCl2, PCl3, CH3Cl. |